MSP Corporation
Magnesium Saves our Planet
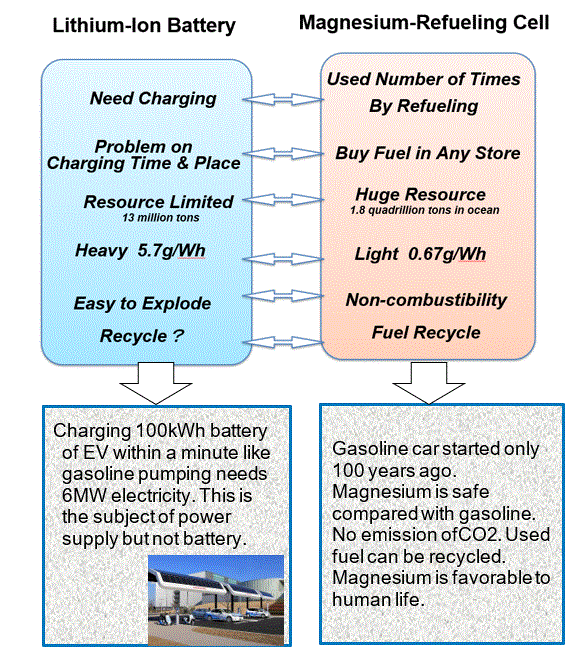
Magnesium Sustainable Power

Difficulty in Magnesium Battery
When magnesium reacts as battery, reaction product shown in left photo is produced on the magnesium surface. Since these reaction residue is is close to insulator, the electric resistance increases and inner part of magnesium covered by insulator cannot react.
Solution
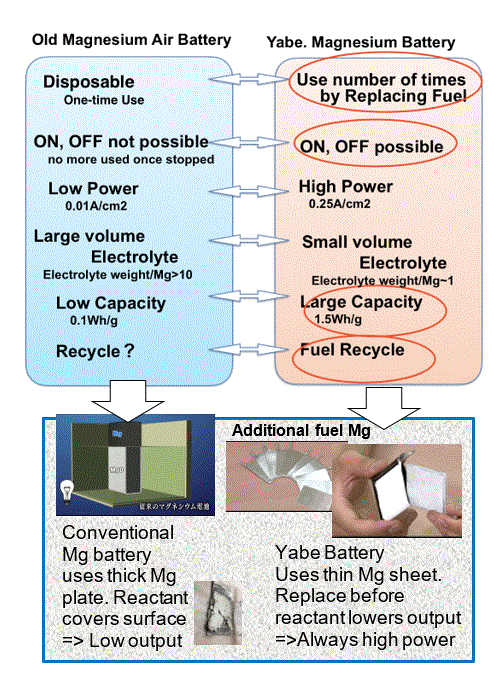
As left bottom figure shows, thick magnesium might be used for a long hours, however, insulating reaction residue is formed on the surface and it inhibits further reaction and output is strongly reduced.
One solution might be to include some chemical component in electrolyte. In this case, however, electrolyte volume increases and electric resistance increase, lowering output power. Furthermore, melting insulator into electrolyte should also increase resistance.
Yabe proposed completely different approach. As left bottom figure shows, magnesium as thin as possible is used and reaction ends before insulator inhibits the reaction and then another magnesium is fed into the system number of times.
Then we succeeded to keep high power output. The other merit is the required electrolyte becomes very small and the distance between two electrodes decreased leading to small resistance.

After magnesium fully reacts, it becomes powder. This powder is taken away enclosed in cloth bag.


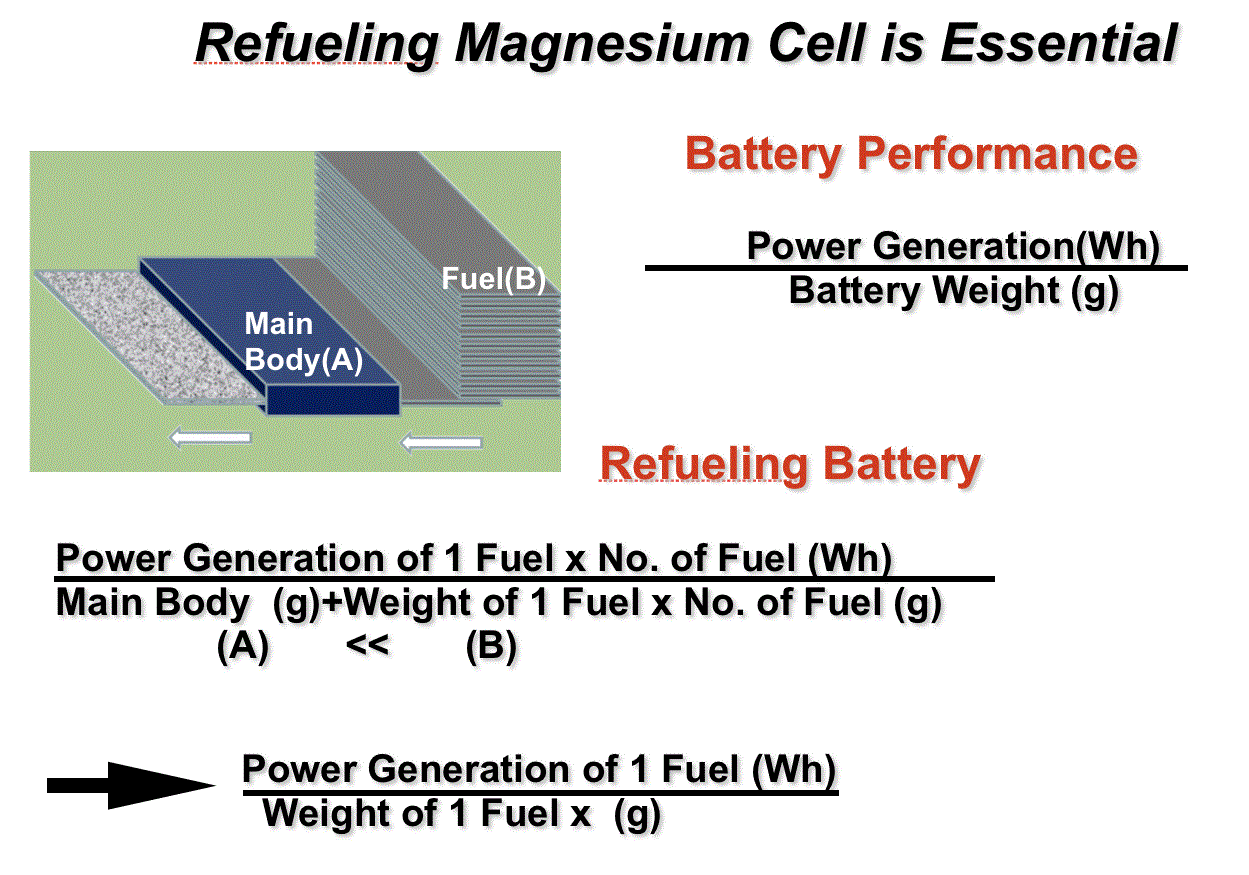
Yabe battery is essentially refueling battery. In general, battery performance is measured by Wh/g which is output/battery weight. In refueling battery, if the amount of fuel is much larger than battery main body, battery weight is negligible and hence performance is determined only by fuel performance without battery body. Thus getting maximum performance of magnesium battery, Yabe battery is essential.
Superiority of Yabe Magnesium Battery